
John Hart, Ph.D., a key collaborator on this study from the Health Science Center, explains: “By using X-ray crystallography and computational methods we were able to precisely pinpoint how the drug interacts with the critical protein in one Schistosome species, and to identify the key differences in this protein in the related parasite species. The research raises hope that this drug can be redesigned to kill the two major schistosome species responsible for 99 percent of schistosomiasis cases worldwide. The drug oxamniquine, the subject of this research, kills just one of the three species of schistosome parasite that infect humans. The problem is that there are very few effective drugs available. No vaccine exists for schistosomiasis, so this disease is controlled using drug treatment, which must be repeated periodically because people become reinfected through contact with water where the intermediate snail hosts live. “We don’t know the death toll from these parasites,” says LoVerde, “but our best estimate is that more than 200,000 people die every year from this infection in Africa alone, making this parasitic disease second only to malaria in terms of mortality.” Welch Foundation – appears in the November 21 online edition of the journal Science.Īdult schistosome parasites are half an inch long and live in the human blood vessels, laying thousands of eggs, many of which become lodged in the liver or bladder wall, causing portal hypertension, liver failure and bladder cancer. 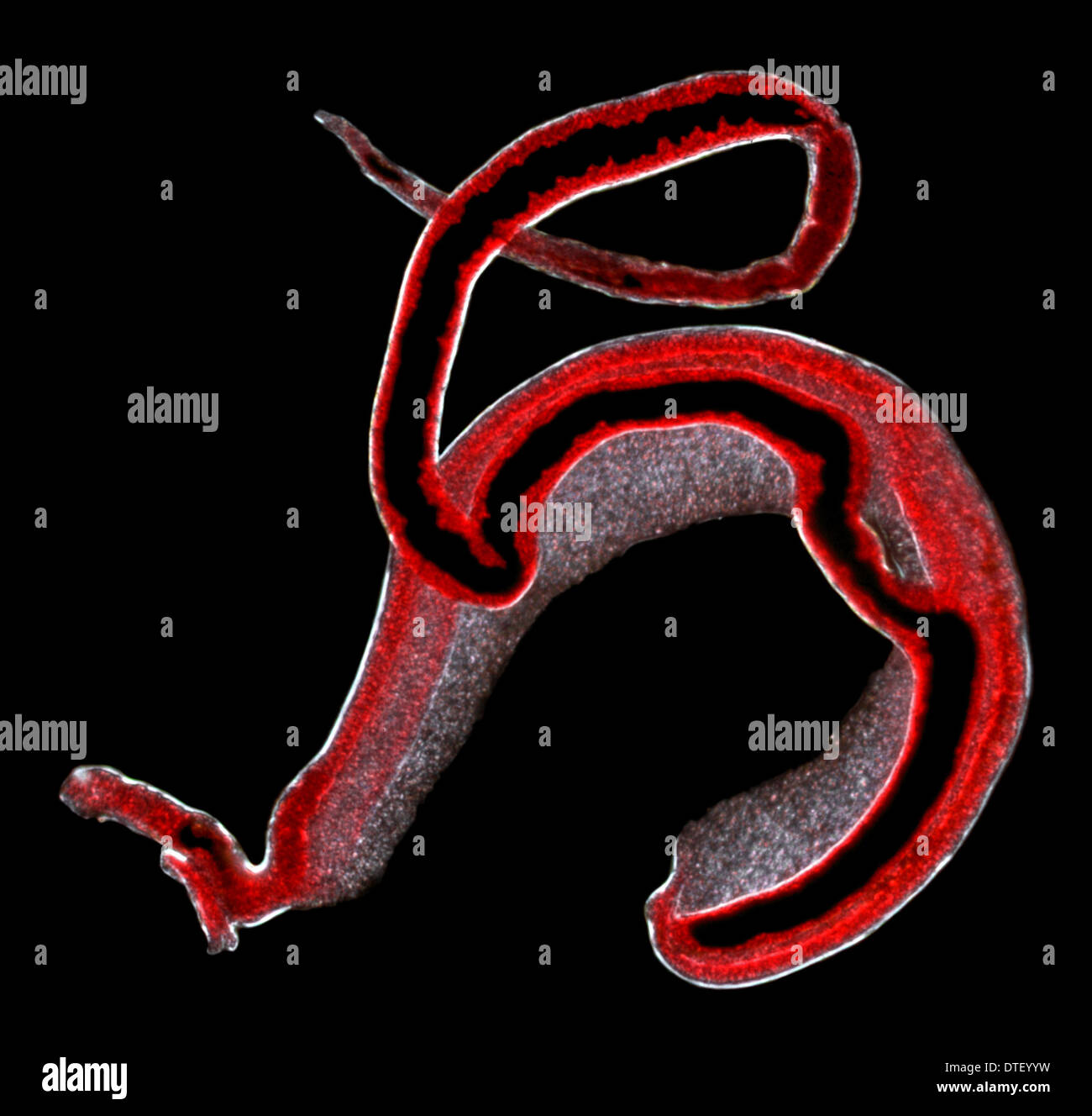 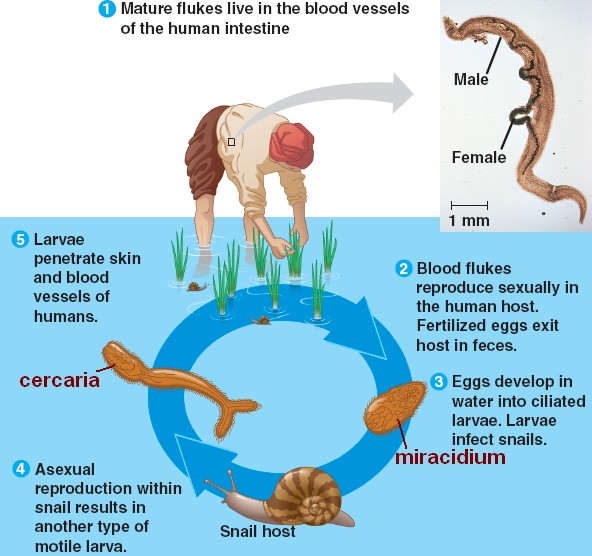
The new study – funded by the National Institutes of Health (NIH), the World Health Organization, the Wellcome Trust and the Robert A. Without the sequence, the work would have been painfully slow.” “The schistosome genome was published in 2009. “This really shows the utility of genome sequencing,” says Anderson. This method is commonly used for fruit flies and other laboratory organisms, but has not previously been possible for schistosome parasites.” “We were able to identify the critical gene by crossing resistant and sensitive worms in the laboratory and then analyzing the genomes of the progeny. “This is a major advance,” said Claudia Valentim, Ph.D., the primary author of the report, who worked at both institutions. The new finding allows detailed understanding of the drugs’ mechanism of action and raises prospects of improved therapies. An international group of scientists led by Tim Anderson Ph.D., at the Texas Biomedical Research Institute and Philip LoVerde Ph.D., at the University of Texas Health Science Center at San Antonio has identified the mutations that result in drug resistance in a parasite infecting 187 million people in South America, Africa and Asia.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
